Courtney Scales DipVN, NCert(Anaesth), RVN – Clinical Educator (Anaesthesia)
Keith Simpson BVSc, MRCVS, MIET(Electronics) – Clinical Director
Joel Huey BA (Hons) – Content Editor
This is Part 2 - Click here to read part 1
Troubleshooting abnormal readings
There may be certain situations in a healthy patient receiving a high FiO2 where the reading is erroneously low, which is more likely than an erroneously high reading (Haskins, 2015).
An erroneously low reading may occur for the following reasons:

• The administration of an alpha-2 agonist (e.g., medetomidine) causes peripheral vasoconstriction, resulting in limited pulsatile blood flow available to obtain a reading. It usually occurs in the early stages of premedication and will eventually wane.
• If the patient is vasoconstricted, then it is more difficult for the probe to read pulsatile blood flow within the vessel. This may occur in hypothermic patients and actively warming the patient can improve blood flow to the periphery.
• Hypotension can reduce blood flow to the capillary bed and readings may be difficult to obtain. Measures to correct the blood pressure should be taken.
• Optical interference from room light can reduce the ability of the probe’s red and infrared light to differentiate the oxyhaemoglobin from deoxyhaemoglobin. The saturation level usually reduces to around 80%. Covering the probe with a drape or blanket helps to block out the room light, as does ensuring the receiver side of the probe is sitting snug against the tissue.
• Some transmission probes may have a tight clip that compresses and pushes the blood from the capillary bed, which is commonly seen when the tissue is thin, like little cat tongues. The probe can have its clamp loosened, wedged open (a syringe cap helps) or taped “open”. The use of a swab placed between the probe can help to align the sides of the probe and reduce the compression of the clip, but if the swab is wet it may make the tongue cold and cause vasoconstriction
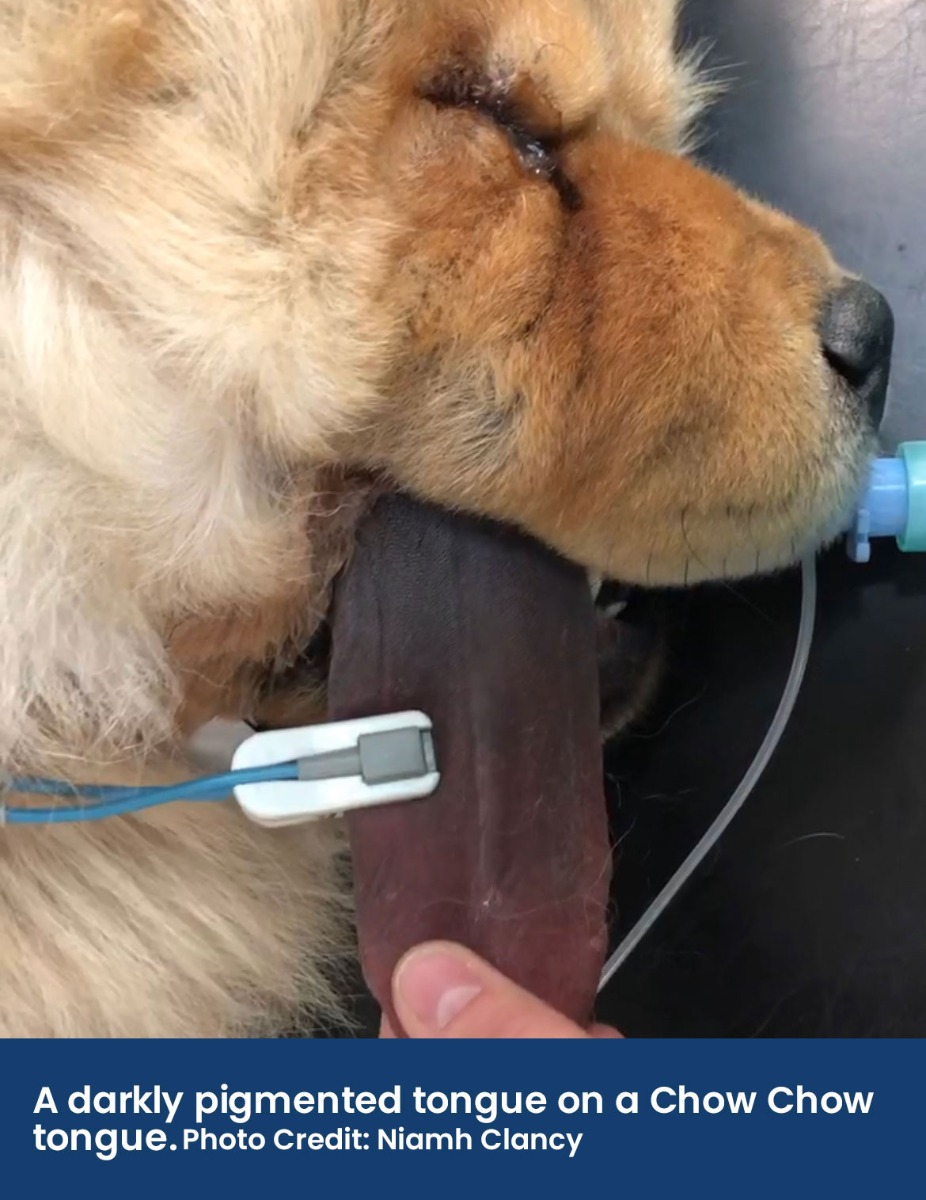
• The tissue where the probe is placed should be pigment and fur-free; the tongue, lip, prepuce, vulva, pinna, or toe webbing can be used. If the patient has pigmented skin (e.g., a dark coloured tongue on a Chow Chow), then this can interfere with the light absorption techniques the pulse oximeter uses.
• Regional circulatory failure can occur if the SpO2 probe is on the same limb or extremity as a non-invasive blood pressure cuff, which is periodically inflated to occlude blood flow.
• In conscious animals, patient movement makes it difficult for the pulse oximetry probe to sense pulsatile arterial blood flow and a reading may not be provided at all.
• Pulse oximetry reads pulsatile arterial blood flow, however there are some instances where there is venous congestion in various organs which can lead to venous pulsation. These readings will be artificially lower as the venous blood has less oxygen in it, so this should be kept in mind when monitoring patients with left-sided heart failure or hepatic congestion.
Why might a patient become hypoxaemic?
Hypoxaemia can occur as a result of reduced oxygen delivery to the patient or due to the patient’s disease process that impairs the diffusion of the oxygen into the alveoli or the ability to carry the oxygen. Hypoxaemia during anaesthesia may occur for the following reasons:
• A low FiO2 is being delivered to the patient. This may be due to an insufficient fresh gas flow supply, a breathing system that is not correctly assembled, a kinked or long endotracheal tube, an exhausted oxygen supply or with the use of nitrous oxide.
• The patient may hypoventilate due to the respiratory depressive side effects that are caused by the anaesthesia drugs administered. Assessing and potentially decreasing the anaesthetic depth, changing patient positioning or supplementing breaths through Positive Pressure Ventilation should also be considered.
• Pulmonary disease can cause ventilation/perfusion (V/Q) mismatch or atelectasis. This is when there is a problem with how oxygen diffuses from the alveoli into the capillaries that surround them. In cases of V/Q mismatch, not all alveoli are being perfused or not all perfused alveoli are being ventilated. It can occur in cases of atelectasis, pneumonia, airway disease and fluid overload.
Using Pulse Oximetry in the Pre and Post Anaesthetic Period
Pulse oximetry is widely used in the peri-anaesthetic period, but it can also be used in the pre- and post-anaesthesia period.
The Pre-Anaesthesia Period
A SpO2 reading prior to the anaesthesia can help guide the expected value in the recovery period. For example, brachycephalic breeds are often chronically hypoxic so knowing “their normal” SpO2 prior to anaesthesia can manage expectations postoperatively.
The Post-Anaesthesia Period
If the patient is experiencing a delayed recovery, consider taking a SpO2 reading and providing supplemental oxygen. A patient may become hypoxaemic in the recovery period for several reasons, including:
• Hypoventilation from residual respiratory side effects of anaesthesia drugs once the patient is transitioned from a high FiO2 to room air, hypothermia or in patients that have increased intracranial pressure.
• Airway obstruction or airway disease can impact the delivery of oxygen to the patient’s lungs and could occur in brachycephalic breeds, cats suffering from feline asthma or patients with Acute Respiratory Distress Syndrome (ARDS).
• In patients with a diffusion impairment or ventilation/perfusion (V/Q) mismatch.
• Atelectasis, or the collapse of parts of the lung, commonly occurs under anaesthesia because of patient positioning and because of the delivery of a high concentration of oxygen under anaesthesia. It is usually self-limiting in the healthy patient and reverses after several hours during recovery.
• A normal SpO2 reading does not always indicate adequate oxygen delivery to the tissue
• For there to be noticeable changes in the SpO2, the PaO2 must fall to a dangerously low level
• It provides an audible and visual pulse rate to the anaesthetist and theatre team
The M880B offers a compact handheld monitor that can be used as a capnograph and pulse oximeter, measuring CO2 and SPO2 signals in patients. Boasting a 4.3” auto-rotating touchscreen for ultra-clear readings, and a battery life of up to 18 hours, this monitor is ideal for accurate readings in any environment.
NEW Suntech Range
The NEW Suntech Vet30 & Vet30E can also be used as a pulse Oximeter. The Suntech range has been designed for BP measurement, but the more advanced options offer pulse oximetry and temperature measurement on top of this. Although still portable, the Suntechs are heavier and not as handheld as the previous monitors, these are designed to be more robust, with a rubber protective case and a flat base for sturdy positioning. The Suntech VET30E is suitable for large animals.
Multi-Parameter Monitors
Burtons larger multi-parameter monitors offer a more accurate way of measuring SpO2. Providing all required data shown on a clear screen so that all the results can be interpreted together. Some monitors even provide extras such as signal strength ratings and recognitions. As mentioned above, the Lightning Multi-Parameter Monitor offers you a signal strength reading of either A (good) or B (could be better), allowing you to adjust the probe position and try to get the most accurate reading possible.
The popular Mindray uMEC12 and ePM12 both feature an extra large, tilted, clear touchscreen. With easy configuration, you can customise your view to include the exact parameters required for the procedure, again, allowing you to view all data at once for the most accurate interpretation. Both of these monitors can have extra modules added on to provide even more measurement capabilities. The anti-interference SpO2 algorithm provides accurate measurement, even when the patient is mobile.
Download this PDF to test your knowledge on practical pulse oximetry
Click here to upload 30 minutes of CPD via the RCVS 1 CPD Platform
References
Brodbelt, D., Blissitt, K., Hammond, R., Neath, P., Young, L., Pfeiffer, D. and Wood, J. (2008). The risk of death: the Confidential Enquiry into Perioperative Small Animal Fatalities. Veterinary Anaesthesia and Analgesia, 35(5), pp.365-373.
Haskins, S. (2015). Monitoring Anesthetized Patients. In: K. Grimm, L. Lamont, W. Tranquilli, S. Greene and S. Robertson, ed., Veterinary Anesthesia and Analgesia, 5th ed. John Wiley & Sons, p.104.
Klaassen, J. (1999). Reference Values in Veterinary Medicine. Laboratory Medicine, 30(3), pp.194-197.
McNally, E., Robertson, S. and Pablo, L. (2009). Comparison of time to desaturation between preoxygenated and nonpreoxygenated dogs following sedation with acepromazine maleate and morphine and induction of anesthesia with propofol. American Journal of Veterinary Research, 70(11), pp.1333-1338
Schauvliege, S. (2016). Patient Monitoring and Monitoring Equipment. In: T. Duke-Novakovski, M. de Vries and C. Seymour, ed., BSAVA Manual of Canine and Feline Anaesthesia and Analgesia., 3rd ed. Gloucester: John Wiley & Sons, p.83
Shino, H., Otsuka-Yamasaki, Y., Sato, T., Ooi, K., Inanami, O., Sato, R. and Yamasaki, M., 2018. Familial Congenital Methemoglobinemia in Pomeranian Dogs Caused by a Missense Variant in the NADH-Cytochrome B5 Reductase Gene. Journal of Veterinary Internal Medicine, 32(1), pp.165-171


